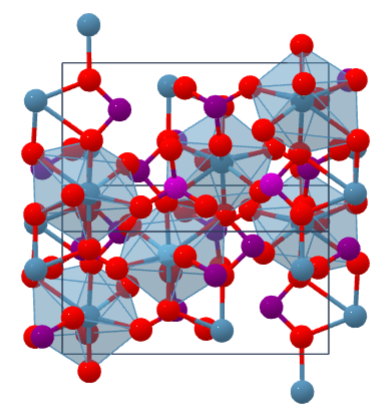
Calcium iodate Monohydrate (Calcium Iodate Monohydrate) with the chemical formula Ca(IO3)2 .H2O [CAS No. 10031-32-0] which in structure monoclinic Its anhydrous form in nature is called Lautarite and its monohydrate form is called Brüggenite.

– Poultry and Livestock Feed :
As an additive to poultry and livestock feed to provide iodine needed for the natural growth of poultry and livestock.
Medication :
– Usage in some ointments and lotions
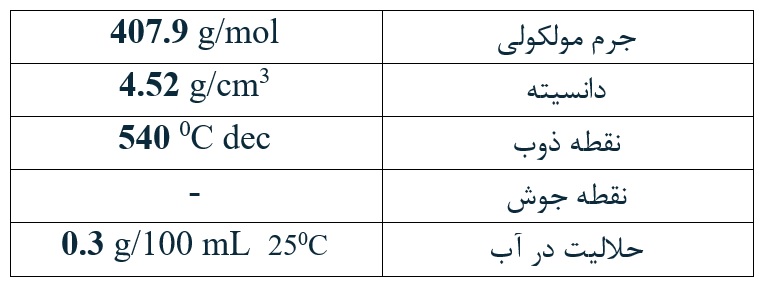
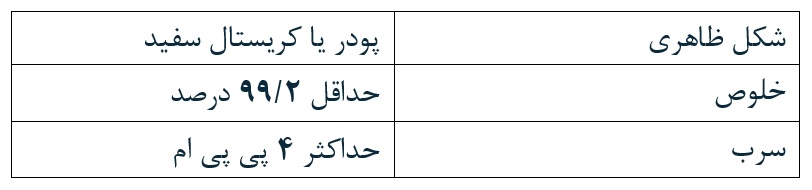

The calcium iodate product of the company has a purity of over 99% and complies with the FCC standard and has a production license from Iran’s Veterinary Organization.
The packaging of all the company’s products is in metallized bags that include 3 layers of polyester, aluminum, and polyethylene.
Safety and Storage :
Calcium iodate may react severely with combustible and reducing agents such as aluminum, organic compounds, hydrogen peroxide, and sulfides. The products resulted from its dangerous decomposition include hydrogen iodide, iodine, and potassium oxides. Avoid storing this material next to the above compounds and store it in an environment with low humidity and away from direct light or heat.
